
The electrons occupy shells or orbits following certain rules : These shells are also denoted by capital letters K,L,M,N,O. As we go away from the nucleus, the numbers go on increasing as 2,3,4,5. The shell nearest to the nucleus is given number 1. The shells or orbits in which electrons revolve are given numbers. The surface generated by the changing plane of the orbit is called a ’shell’. Hence, it was concluded that ’atom is a three dimensional body’. it is a neutral particle.Įxtra-nuclear part electrons and their distributionīohr’s model suggested that there is a nucleus at the center of the atom and electrons revolve around the nucleus in selected stationary orbits. Absolute charge on an electron is - 1.602 x 10−19 C It carries a charge equal in magnitude but opposite in sign to that carried by an electron = 1.602x 10−19 C It carries no charge i.e. Absolute mass of a neutron is 1.676 x 10−24 g = 1.676 x10−27 kg Relative mass of a neutron is 1.009665 u Charge An electron has unit negative charge. Absolute mass of a proton is 1.672 x 10−24g = 1.672 x10−27 kg Relative mass of a proton is 1.007277 u The mass of a neutron is almost equal to that of a proton. Absolute mass of an electron is 9.1x10−28g = 9.1 x10−31 kg Relative mass of an electron is 0.00054859 u The mass of a proton is equal to that of a hydro- gen atom. Property Electron −1e0 Proton +1p0 Neutron 0n1 Mass The mass of an electron is about 1/1836 that of a hydrogen atom. 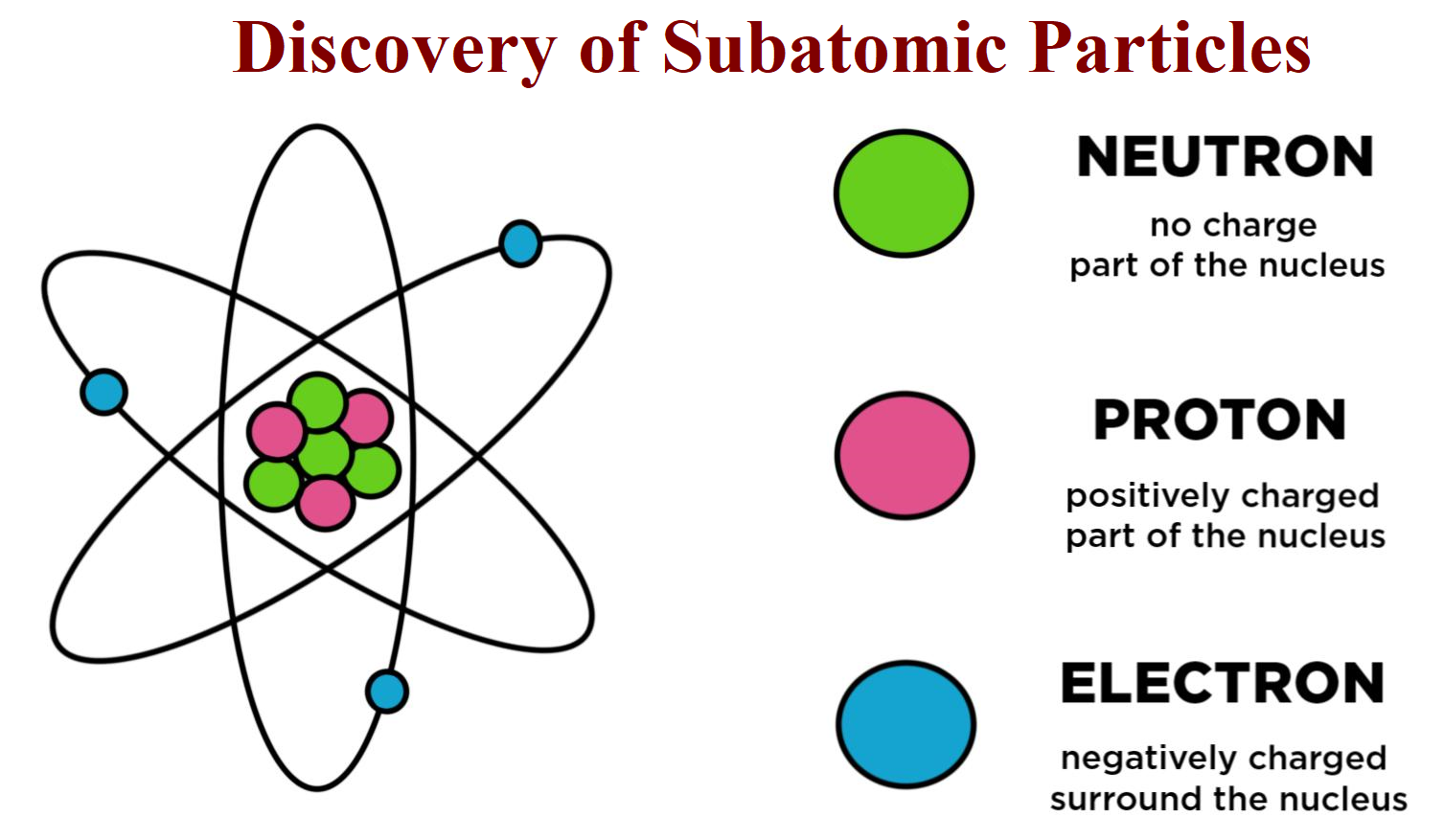
Their characteristic properties are given below.Ĭharacteristic properties of the sub-atomic particles There are three sub–atomic particles –protons, electrons and neutrons. The particles constituting an atom are called sub–atomic particles or elementary particles.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
